Yuflyma strives to tap into the U.S. market
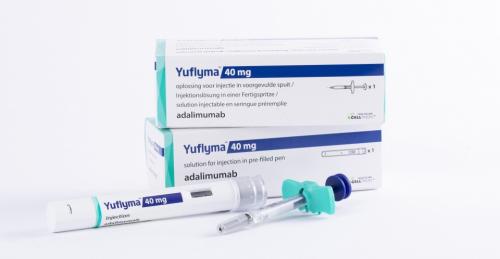
South Korean pharmaceutical company Celltrion said on May 25 that its Humira biosimilar CT-P17, or Yuflyma, had won the approval of the U.S. Food and Drug Administration (FDA).Beginning in July, Celltrion plans to sell Yuflyma, a high-concentration and citrate-free formulation of Humira biosimilar, in the United States.Humira is a monoclonal antibody used to treat such diseases as rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, and ankylosing spondylitis.The FDA gave the nod to Yuflyma for the treatment of eight conditions, including the above-mentioned symptoms.Yuflyma is Celltrion’s fifth biosimilar approved for use in the U.S. The firm is scheduled to offer patients pre-filled syringes and autoinjector administration options.“Yuflyma offers patients a high-concentration and citrate-free formulation of adalimumab biosimilar, providing an alternative treatment option for patients. It represents a key treatment option in patient care and patient choice,” Celltrion USA chief commercial officer Tom Nusbickel said.“As a leading global biopharmaceutical company, we are leveraging our unique heritage in biotechnology, supply chain excellence, and best-in-class sales capabilities to expand the availability of high-quality biosimilars for U.S. patients."Celltrion expected that its new biosimilar would win a substantial portion of the market because more than 80 percent of U.S. patients treated with Humira depended on a high-concentration and citrate-free formulation.The company also noted that it would seek an interchangeability designation from the FDA for Yuflyma, possibly by the end of next year.The designation enables patients to switch back and forth between an interchangeable biosimilar and a reference product, as compared to receiving treatment with just the reference product.To win the designation, a biosimilar has to meet additional requirements outlined by law.The share price of Celltrion went down 3.3 percent on May 25 on the South Korean stock exchange.키워드
이 기사를 공유합니다
Kevin Chung
(jumphigher55@aol.com)

